ZetaView® Subpopulations Size Fluorescence Zeta Potential Concentration Colocalization Next Generation NTA
Applications
EXTRACELLULAR VESICLES
Research on Exacellular Vesicles is thriving. Nanoparticle Tracking Analysis plays an important role in this development due to in vitro measurement of multiple physical parameters such as size, concentration, surface charge, and phenotype characteristics
Viruses
Nanoparticle Tracking Analysis can be used to quantify total concentration of virions, analyse the amount of successfully loaded virions as well as virus aggregatiion.
Biological nanoparticles, such as extracellular vesicles (EVs), exosomes, lipids, viruses or proteins can be found in almost all fluids of living tissue. The Particle Metrix ZetaView® Nanoparticle Tracking Analysis technology is capable to characterize them reliably in water or physiological buffers. You can simultaneously get information about particle size, particle concentration and surface charge with just one single measurement of a particle sample. Similarly, cluster analysis based on similar properties is possible.
The x30 Series at a Glance
For more information on the ZetaView® or to discuss your requirements, please contact us.
Upcoming Events
Get the news
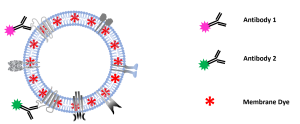
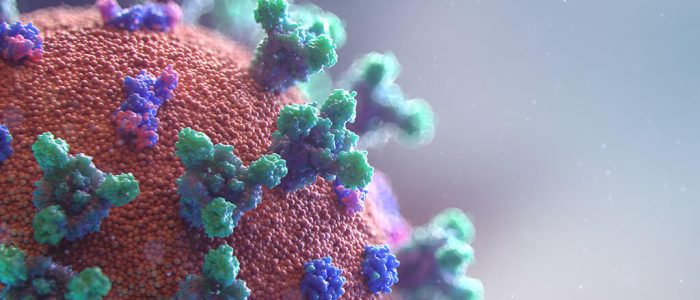
Colocalization-NTA of multi-stained platelet derived MSC-EV preparations
After successfully showing C-NTA of double-stained MSC-EVs in our previous study (https://bit.ly/43GG0Yk) and confirming
the results with image flow cytometry, this note describes C-NTA of multi-stained MSC-EVs using a PMX-430 QUATT NTA
system with four lasers.
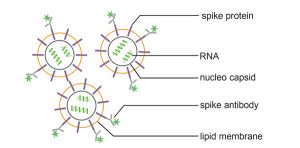
Specific detection of SARS-CoV-2 corona virus from cell culture supernatants by fluorescence-based Nanoparticle Tracking Analysis
In this new application note we describe a quick determination of the physical titer of coronaviruses by fluorescence-based Nanoparticle Tracking Analysis and specific spike antibody recognition.
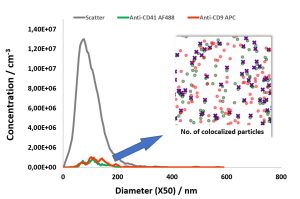
NTA goes colocalization:
Detection and measurement of CD9 and CD41 on
double-stained human platelet derived MSC-EV
preparations
In this report, we describe the quantification of colocalization ratios on double-stained MSC-derived EVs using fluorochrome conjugated antibodies against the
cell surface antigens CD9 and CD41, which also serve as EV marker proteins.