
Analysis of 25 nm functionalized QDs using fluorescence-NTA with ZetaView® QUATT system
Application Note Download Abstract While it is uncertain how far the NTA community is able to push the limits of current technology in order to

0x consumables
5x faster switching
10x faster cleaning
12x fluorescence channels
∞x statistics
…and much more!

We measure your products in a measuring range of 0.8 nm to 300 μm, if possible in original concentration
Full recovery of samples measured by a nanoparticle tracking analyzer (NTA) often becomes challenging particularly if the samples are intended for further use after measurement.

Full recovery of samples measured by a nanoparticle tracking analyzer (NTA) often becomes challenging particularly if the samples are intended for further use after measurement. Furthermore, depending on isolation techniques and purification methods, especially for biological nanoparticles, the yield of samples is often very low in terms of volume and concentration. Here, we describe a convenient way to fully recover samples in order to re-use them entirely for other purposes after they have been measured in the Particle Metrix ZetaView® instrument when equipped with an NTA- cell assembly.
The Particle Metrix ZetaView® is widely used in life science research for measuring size, concentration, fluorescence and zeta potential of biological nanoparticles, such as extracellular vesicles, proteins, viruses, prions and other cell-derived nanoparticles (1, 2, 3). Despite well-developed extraction techniques and sophisticated isolation methods, the resulting yield of purified nanoparticles is often very low, particularly if isolated only on the laboratory scale.
For that reason, it is important to sacrifice as small a sample volume as possible for measurements -or even better, to recover the sample once it has been measured.
Using the Particle Metrix ZetaView® in combination with the NTA- cell assembly (not possible with the Z-NTA cell assembly) you can easily recover valuable samples after they have been measured.
Since the NTA- and X-NTA cell assemblies feature a flow-through cell channel system with the inlet and outlet ports located on opposite sides to each other (see Figure 1), you can easily inject your sample on the right side and re-collect it on the left side requiring a minimum of 600µl sample volume.
After the sample has been measured, it can easily be collected on the left outlet of the cell assembly in an Eppendorf tube or similar vial. Figure 2 shows the complete set-up.
After measurement, the sample is not lost and can be re-collected in an appropriate reaction tube. With this set-up, the sample flow is unidirectional from right to left because the check valves do not allow re-collecting the sample back by drawing it into the syringe.
To have the flexibility to draw the sample back into the syringe after having done the measurement, the check valves (see Figure 1) must be unscrewed in order to allow the sample to go bi-directionally through the flow cell. This set-up is shown in Figure 3.
With the check valves unscrewed from the inlet and outlet ports of the cell assembly, the sample can either be re-collected into a tube at the left side or sucked back into the original syringe at the right side without using the left outlet port. This way of re-collecting the sample is possible if the sample volume that is injected into the instrument does not exceed 1000µl. A larger volume would exceed the capacity of the cell, meaning sample begins to flow out of the left outlet port.
Re-collecting the entire sample either in a tube or by drawing the sample back into the syringe is of great benefit for researchers struggling with high limited sample volume or very low particle concentrations that needs to be used for further experiments. Therefore, sacrificing as little sample as possible, especially if the particle concentration is so low that the sample needs to be measured undiluted, is a great advantage.

Application Note Download Abstract While it is uncertain how far the NTA community is able to push the limits of current technology in order to
After successfully showing C-NTA of double-stained MSC-EVs in our previous study (https://bit.ly/43GG0Yk) and confirming
the results with image flow cytometry, this note describes C-NTA of multi-stained MSC-EVs using a PMX-430 QUATT NTA
system with four lasers.
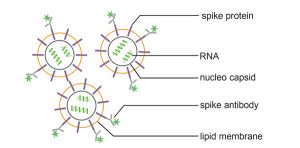
In this new application note we describe a quick determination of the physical titer of coronaviruses by fluorescence-based Nanoparticle Tracking Analysis and specific spike antibody recognition.
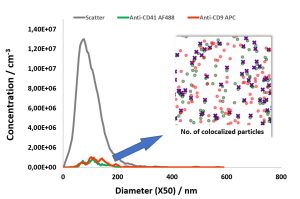
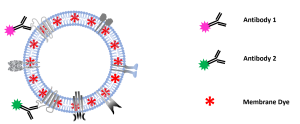
In this report, we describe the quantification of colocalization ratios on double-stained MSC-derived EVs using fluorochrome conjugated antibodies against the
cell surface antigens CD9 and CD41, which also serve as EV marker proteins.

In addition to using Nano Particle Tracking Analysis (NTA) to measurement the size distribution and concentration of EV samples, both Microfluidic Resistive Pulse Sensing (MRPS) and the Single Particle Interference Reflectance Image Sensor (SP-IRIS) methods have been widely used as an alternative means of characterizing EVs. In this note, we relay two specific cases for using the ZetaView® NTA system to achieve relative “high-throughput” analysis of many EV samples, along estimations of throughput for MRPS & SPIRIS methods for an equivalent number of samples; further, we establish realistic estimates for the high cost of ownership for operating MRPS & SP-IRIS systems as a result of the cost of consumables as well as the substantially greater amount of time spent to run the same number of samples.
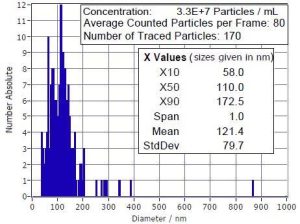
Along with generally accepted methods are some “tricks of the trade” such as the addition of Tween® or BSA; however, some of those additions are problematic.
Headquarter
Support
Opt-out complete; your visits to this website will not be recorded by the Web Analytics tool. Note that if you clear your cookies, delete the opt-out cookie, or if you change computers or Web browsers, you will need to perform the opt-out procedure again.
You may choose to prevent this website from aggregating and analyzing the actions you take here. Doing so will protect your privacy, but will also prevent the owner from learning from your actions and creating a better experience for you and other users.
The tracking opt-out feature requires cookies to be enabled.